Owlstone Medical commences next phase in world’s largest breath-based clinical trial for early cancer detection
Published on 22 Sep 16
NHS funded Lung Cancer Indicator Detection (LuCID) program to recruit up to 3,000 patients across 21 international sites
Cambridge, UK, September 22, 2016: Owlstone Medical, a diagnostics company, has announced that it is expanding its Lung Cancer Indicator Detection (LuCID) clinical trial. The study, originally funded by the Small Business Research Initiative for Healthcare (SBRI Healthcare), an NHS England funded initiative, aims to save 10,000 lives and save the NHS £245 million by 2020 with a quick, easy to use, non-invasive, high-compliance breath test to detect lung cancer. The study will recruit up to 3,000 patients across 21 sites in the UK and Europe, making it the world’s largest breath-based study ever undertaken for early cancer detection.
If detected at stage 1, the 5-year survival rate for lung cancer is 54%, but this drops to just 4% if the cancer is detected at stage 4. Current lung cancer screening tests, such as low dose computed tomography (CT screening), have been shown to result in a high number of false-positives and therefore unnecessary, invasive follow-up procedures on healthy patients.
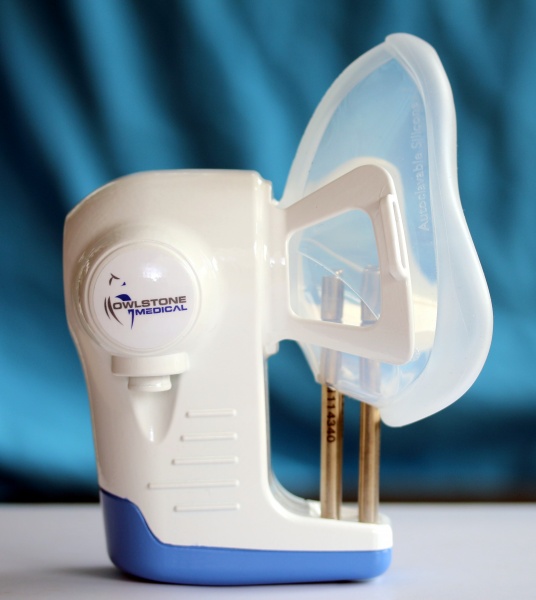
The mutations that drive the development of lung cancer have downstream effects on the metabolites we exhale, even in the very early stages of lung cancer. Owlstone Medical uses the Respiration Collector for In Vitro Analysis (ReCIVA ) , in combination with the Field Asymmetric Ion Mobility Spectrometer (FAIMS) sensor platform, to accurately and selectively detect lung cancer volatile organic compounds (VOCs) in breath.
The trial is moving to the next phase as a consequence of positive interim sensitivity and specificity data, when compared to other available tests, with over 97% of patients reporting the breath test to be comfortable to perform.
The study is being led by Dr Robert Rintoul, lead clinician for Thoracic Oncology and Director of the Clinical Trials Unit at Papworth Hospital, Cambridge, UK, and will recruit patients in order to validate breath biomarkers for the early detection of cancer and differentiation between benign and malignant tumors, which is a major problem in current screening approaches. The trial will compare Owlstone Medical’s platform with current CT and PET screening tests, with the aim of demonstrating its ability to reduce the number of false-positive results and complications seen from invasive biopsy follow-ups.
Patient recruitment is on target with twelve sites in the UK and elsewhere in Europe currently recruiting with nine more sites to come on board shortly.
Commenting on the study, Dr Robert Rintoul said: “The need to explore novel approaches for the early detection in lung cancer has never been greater. I am particularly excited about the opportunity that the Owlstone LuCID project brings to study the role of VOCs for the early detection of lung cancer. If VOCs work, then it may be possible to use a simple breath test to help identify whether someone has lung cancer or not.”
Billy Boyle, co-founder and CEO at Owlstone Medical, said: “Early detection through improved screening is the greatest opportunity for improving patient outcomes. Owlstone Medical was created specifically to advance our breathalyzer for very early stage diagnosis of disease, to enable more effective treatment and better patient outcomes.”
At the European Respiratory Society International Congress in London earlier this month, the academic steering committee for Owlstone’s LuCID clinical trial Phase II project met, and Owlstone held a workshop for its Stratification of Asthma Treatment by Breath Analysis (STRATA) SBRI Healthcare Phase I project, to combine input of a wide range of stakeholders into the project and report initial trial progress. This project aims to allow patients suffering from difficult-to-treat severe asthma to quickly find the treatment that will work best for them, increasing quality of life for patients and reducing health care costs.
To find out more about Owlstone Medical visit: www.owlstonemedical.com
Notes to Editors
Image of Owlstone Medical’s Respiration Collector for In Vitro Analysis (ReCIVA)
For a high resolution image please contact lorna.cuddon@zymecommunications.com
Media contact:
For more information please contact:
Lorna Cuddon, Zyme Communications
lorna.cuddon@zymecommunications.com
+44 (0)7811 996 942
About Owlstone Medical:
Owlstone Medical has developed a breathalyzer for disease. With a focus on non-invasive diagnostics for cancer, inflammatory disease and infectious disease, the company aims to save 100,000 lives and $1.5B in health care costs by 2020.
The company’s microchip FAIMS (Field Asymmetric Ion Mobility Spectrometer) sensor is a platform technology that can be programmed in software to detect targeted biomarkers of disease in breath (and other bodily fluids). FAIMS can be used in clinical diagnostics and precision medicine with application in cancer and a wide range of other medical conditions. Highly sensitive and selective, these tests allow for early diagnosis when treatments are more effective and where more lives can be saved.
Owlstone Medical is currently developing tests for lung and colorectal cancer, two of the most common cancer killers worldwide, and for asthma stratification by therapeutic response. The company also sells R&D tools and services to academic, clinical and pharma partners who want to develop breath based diagnostics for their own applications.
Founded in 2004 as a spin-out from the Engineering Department at the University of Cambridge, Owlstone Inc is a profitable business selling its proven FAIMS technology to military and industrial customers globally. Owlstone Medical was spun out from Owlstone Inc in 2016 to develop and commercialise FAIMS in diagnostic applications.
Owlstone Medical is headquartered in Cambridge, UK, with offices in London, UK